|
The experimental results were very different. The Thomson model had predicted a small deviation of a few degrees as the positive and negative charges are distributed throughout the whole atom in this model. (iii) An extremely small number of very few α-particles deviated through large angles, and about one in a million α-particles retraced their path. (ii) A small number of α-particles underwent small deviation. 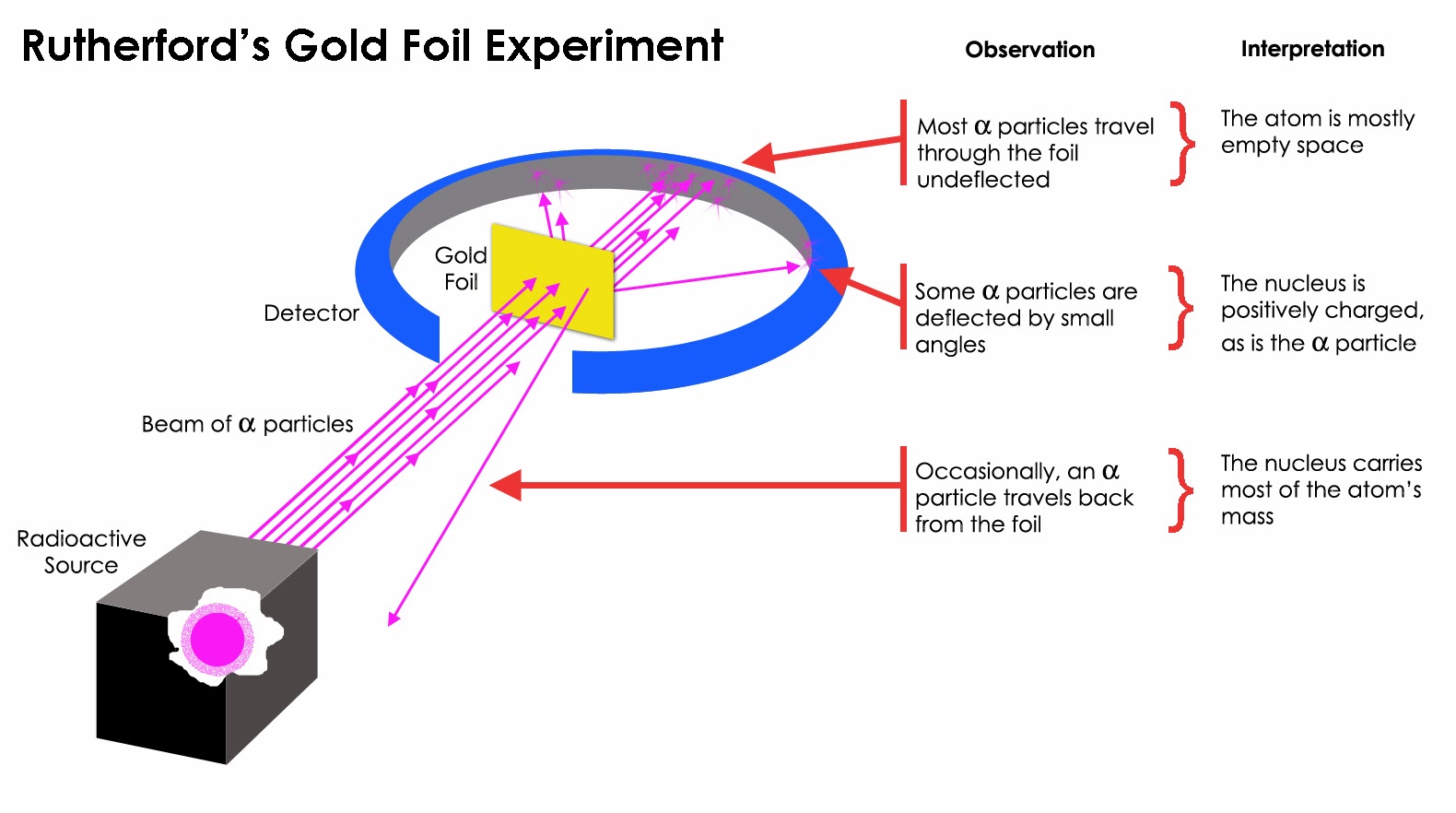
(i) Most of the α-particles passed undeviated. Two students working in Rutherford’s laboratory, Geiger & Marsden, bombarded highly energetic α-particles onto a thin Gold foil. In subsequent sections we will study the Rutherford and Bohr models of the atom. Several models were proposed to explain atomic structure : the Thomson model, the Rutherford model, the Bohr model and so on.  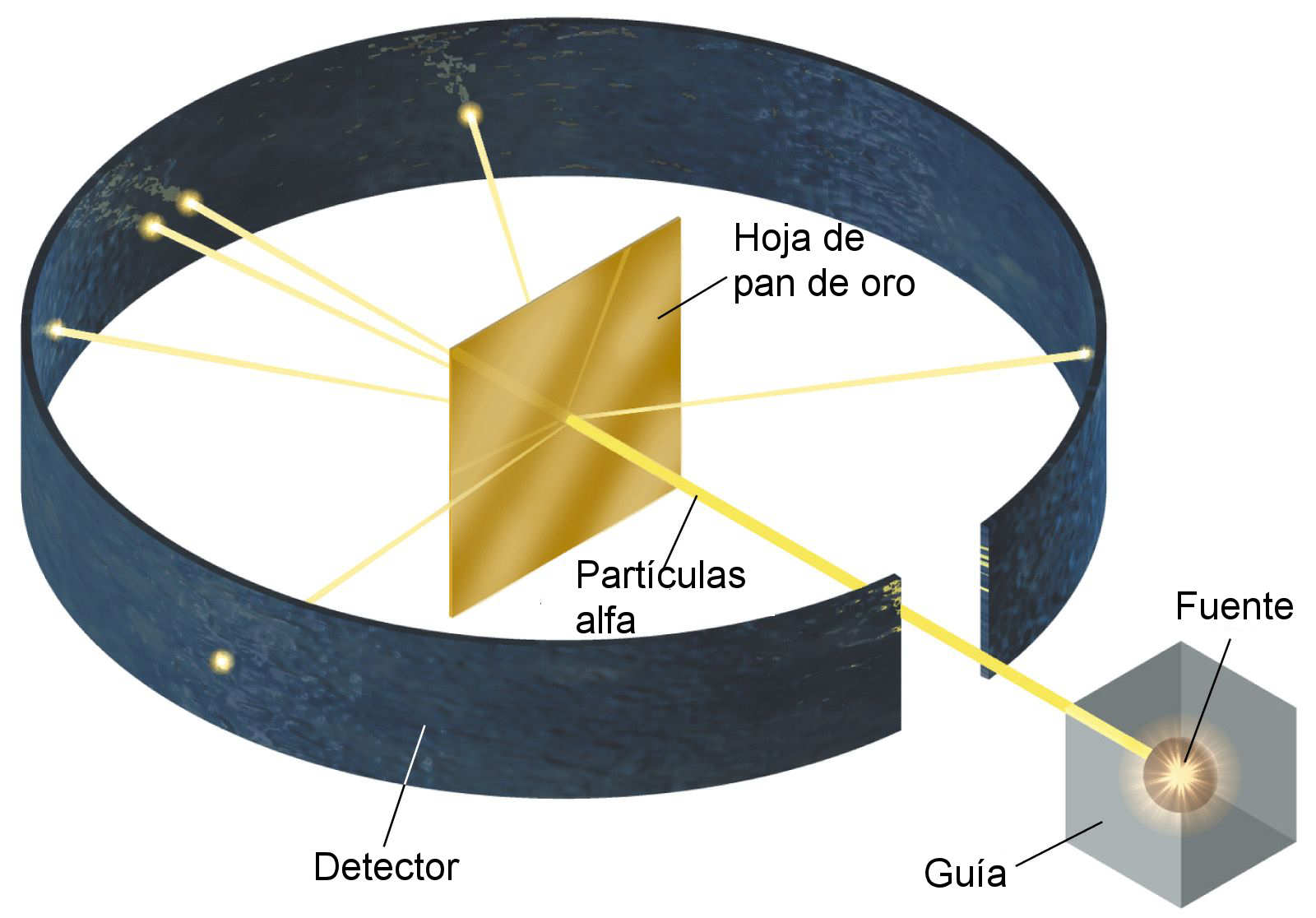
The exact structure of the atom and the forces responsible for holding these particles together were the subject of investigation in those times. Until the end of the 19th century, it was believed that atoms were indivisible, but later, it was found that atoms are consisted of smaller subatomic particles-electrons, protons and neutrons.Įlectrons are light, negatively charged particles while protons are heavy, positively charged particles and neutrons are heavy, neutral particles. Matter consists of small particles called molecules which again consist of atoms.
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
